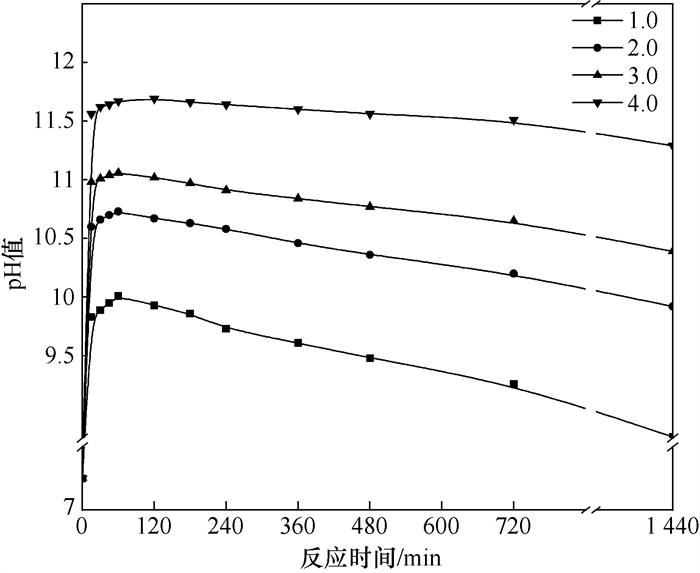
Variation of pH value of leaching solution during steel slag dissolution
-
摘要: 为了掌握钢渣堆存时对环境造成的影响,本文研究了钢渣在溶出过程中浸出液pH值的变化规律,分析了钢渣碱度、氧化镁质量分数、水温、钢渣粒度、液固比等因素对钢渣浸出液pH值的影响规律,并与现场钢渣进行了对比试验。结果表明:钢渣溶出后浸出液pH值在10~12,随时间呈先升高后降低的趋势;提高钢渣碱度及氧化镁质量分数均使浸出液的pH值升高;水温的升高、钢渣粒度和液固比的增大可降低浸出液的pH值。Abstract: This study explored the change law changing pattern of pH value of steel slag leaching solution in the process of steel slag dissolution with an aim to study the influence of steel slag on environment.Specifically, it analyzed the influence law of steel slag basicity, magnesium oxide content, water temperature, steel slag particle size, liquid-solid ratio and other factors on pH value of steel slag leaching solution and conducted a comparative test with on-site steel slag.Results show that the pH value of the leaching solution after steel slag dissolution is in the range of 10~12, and with the change of time, the pH value first increases and then decreases.Increasing the basicity of steel slag and the content of magnesium oxide can increase the pH value of the leaching solution.The increase of water temperature, steel slag size and liquid-solid ratio can reduce the pH value of the leaching solution.
-
Key words:
- steel slag /
- leach liquor /
- pH value /
- alkalinity /
- magnesium oxide content
-
表 1 现场钢渣的成分组成质量分数
Table 1. Composition mass fraction of steel slag in the field
% CaO SiO2 Fe2O3 Al2O3 P2O5 MgO 其他 48.13 13.63 25.19 1.99 2.72 3.82 4.52 表 2 不同碱度渣样的成分组成质量分数
Table 2. Composition of slag samples with different alkalinity
% 渣样碱度 CaO SiO2 Fe2O3 MgO Al2O3 P2O5 R1.0 30.0 30.0 27.0 6.0 2.0 5.0 R2.0 40.0 20.0 27.0 6.0 2.0 5.0 R3.0 45.0 15.0 27.0 6.0 2.0 5.0 R4.0 48.0 12.0 27.0 6.0 2.0 5.0 表 3 不同MgO含量渣样的成分组成质量分数
Table 3. Composition of slag samples with different MgO contents
% CaO SiO2 Fe2O3 Al2O3 P2O5 MgO 51.2 12.8 27.0 2.0 5.0 2.0 49.6 12.4 27.0 2.0 5.0 4.0 48.0 12.0 27.0 2.0 5.0 6.0 46.4 11.6 27.0 2.0 5.0 8.0 44.8 11.2 27.0 2.0 5.0 10.0 表 4 图 3中钢渣不同位置能谱分析结果
Table 4. Energy spectrum analytic results of steel slag at different positions in Fig. 3
% 元素 Ca Mg Fe Al P Si O 浸出前 点位1 43.12 3.56 14.07 0.84 8.55 2.18 23.85 点位2 44.43 4.49 17.04 0.92 8.03 2.43 19.86 浸出后 点位1 23.43 1.17 11.51 0.54 7.72 2.05 49.08 点位2 23.67 1.54 13.54 0.66 7.69 1.97 47.67 -
[1] 辜海芳. 2021年我国钢铁行业运行情况分析[J]. 冶金经济与管理, 2022(3): 18-21. https://www.cnki.com.cn/Article/CJFDTOTAL-YJJJ202203005.htmGu Haifang. Analysis on the status of China iron and steel industry in 2021[J]. Metallurgical Economy and Management, 2022(3): 18-21. https://www.cnki.com.cn/Article/CJFDTOTAL-YJJJ202203005.htm [2] 赵立杰, 张芳. 钢渣资源综合利用及发展前景展望[J]. 材料导报, 2020, 34(S2): 1319-1322, 1333. https://www.cnki.com.cn/Article/CJFDTOTAL-CLDB2020S2065.htmZhao Lijie, Zhang Fang. Comprehensive utilization and development prospect of steel slag resources full text replacement[J]. Materials Reports, 2020, 34(S2): 1319-1322, 1333. https://www.cnki.com.cn/Article/CJFDTOTAL-CLDB2020S2065.htm [3] 王淑娟. 钢渣的利用现状及发展趋势分析[J]. 黑龙江科学, 2019, 10(2): 160-161. https://www.cnki.com.cn/Article/CJFDTOTAL-HELJ201902071.htmWang Shujuan. Analysis on the utilization status and development trend of steel slag[J]. Heilongjiang Science, 2019, 10(2): 160-161. https://www.cnki.com.cn/Article/CJFDTOTAL-HELJ201902071.htm [4] 王吉凤, 付恒毅, 闫晓彤, 等. 钢渣综合利用研究现状[J]. 中国有色冶金, 2021, 50(6): 77-82. https://www.cnki.com.cn/Article/CJFDTOTAL-YSYL202106015.htmWang Jifeng, Fu Hengyi, Yan Xiaotong, et al. Research status of comprehensive utilization of steel slag[J]. China Nonferrous Metallurgy, 2021, 50(6): 77-82. https://www.cnki.com.cn/Article/CJFDTOTAL-YSYL202106015.htm [5] 孔志云, 黄纬斌, 杜亚杰, 等. 钢渣堆积过程中重金属浸出物研究[J]. 中国金属通报, 2020(11): 108-109. https://www.cnki.com.cn/Article/CJFDTOTAL-JSTB202011055.htmKong Zhiyun, Huang Weibin, Du Yajie, et al. Study on heavy metal extract in steel slag accumulation process[J]. China Metal Bulletin, 2020(11): 108-109. https://www.cnki.com.cn/Article/CJFDTOTAL-JSTB202011055.htm [6] 朱斌鹏. 冶金固废资源化利用现状及发展[J]. 世界有色金属, 2021(23): 7-9. https://www.cnki.com.cn/Article/CJFDTOTAL-COLO202123003.htmZhu Binpeng. Present situation and development of resource utilization of metallurgical solid waste[J]. World Nonferrous Metals, 2021(23): 7-9. https://www.cnki.com.cn/Article/CJFDTOTAL-COLO202123003.htm [7] 李亚东, 徐征, 张汉平, 等. 综合利用视角下冶金工业固体废物钢渣的处理[J]. 化工设计通讯, 2021, 47(12): 149-150. https://www.cnki.com.cn/Article/CJFDTOTAL-WGTX202112074.htmLi Yadong, Xu Zheng, Zhang Hanping, et al. Treatment of steel slag from the perspective of comprehensive utilization[J]. Chemical Engineering Design Communications, 2021, 47(12): 149-150. https://www.cnki.com.cn/Article/CJFDTOTAL-WGTX202112074.htm [8] 刘瑛, 方宏辉, 卢丽君. 钢渣处理与综合利用技术研究进展[J]. 化学工程与装备, 2014(9): 190-192. https://www.cnki.com.cn/Article/CJFDTOTAL-FJHG201409061.htmLiu Ying, Fang Honghui, Lu Lijun. Research progress on treatment and comprehensive utilization of steel slag[J]. Chemical Engineering & Equipment, 2014(9): 190-192. https://www.cnki.com.cn/Article/CJFDTOTAL-FJHG201409061.htm [9] Zhu Z Q, Gao X, Ueda S, et al. Contribution of mineralogical phases on alkaline dissolution behavior of steelmaking slag[J]. ISIJ International, 2019, 59(10): 1908-1916. doi: 10.2355/isijinternational.ISIJINT-2019-049 [10] Zhu Z Q, Gao X, Ueda S, et al. Alkali elution of various mineralogical phases in steelmaking slag[C]//Rewas 2019, The Minerals, Metals & Materials Series, 2019: 215-220. [11] Ruan F, Kawanishi S, Sukenaga S, et al. Effect of the silicate structure on calcium elution behaviors of calcium-silicate based mineral ases in aqueous solution[J]. ISIJ International, 2020, 60(3): 419-425. doi: 10.2355/isijinternational.ISIJINT-2019-263 [12] 李宏杰, 吕明利, 刘莲云, 等. 钢渣在纯水与海水中的溶出行为[C]//冶金研究. 北京: 北京科技大学, 2007: 395-397. [13] 刘英金, 宋波, 毛璟红, 等. 钢渣中钙浸出的实验研究[C]//2008年全国冶金物理化学学术会议专辑(上册). 北京: 中国稀土学报, 2008: 94-97. [14] 彭犇, 王晟, 邱桂博, 等. 钢渣处理技术现况和探讨[J]. 工业加热, 2018, 47(6): 55-57. https://www.cnki.com.cn/Article/CJFDTOTAL-GYJR201806017.htmPeng Ben, Wang Sheng, Qiu Guibo, et al. Current situation and discussion of steel slag process technology[J]. Industrial Heating, 2018, 47(6): 55-57. https://www.cnki.com.cn/Article/CJFDTOTAL-GYJR201806017.htm [15] 王帅, 谢丽, 盛杰, 等. 钢渣在环境治理中的应用及其研究进展[J]. 工业水处理, 2008, 28(12): 14-18. https://www.cnki.com.cn/Article/CJFDTOTAL-GYSC200812004.htmWang Shuai, Xie Li, Sheng Jie, et al. Application of steel slag to the environmental pollution treatment and its research progress[J]. Industrial Water Treatment, 2008, 28(12): 14-18. https://www.cnki.com.cn/Article/CJFDTOTAL-GYSC200812004.htm [16] 饶磊. 转炉钢渣成分、结构及性能间内在规律及其应用研究[D]. 北京: 北京科技大学, 2020. [17] 李继铮, 方宏辉, 王悦, 等. 不同处理方式的钢渣显微结构特征分析[J]. 河南建材, 2009(6): 78-79. https://www.cnki.com.cn/Article/CJFDTOTAL-HNJC200906047.htmLi Jizheng, Fang Honghui, Wang Yue, et al. Analysis of microstructure characteristics of steel slag treated with different methods[J]. Henan Building Materials, 2009(6): 78-79. https://www.cnki.com.cn/Article/CJFDTOTAL-HNJC200906047.htm [18] Guthrie G. Geochemical aspects of the carbonation of magnesium silicates in an aqueous medium[C]//Office of Scientific & Technical Information Technical Reports. Los Alamos National Laboratory, 2001. [19] 王强. 钢渣的胶凝性能及在复合胶凝材料水化硬化过程中的作用[D]. 北京: 清华大学, 2010. [20] Tai C Y, Chen W R, Shih S M. Factors affecting wollastonite carbonation under CO2 supercritical conditions[J]. AIChEJournal, 2006, 52(1): 292-299. doi: 10.1002/aic.10572 [21] Huijgen W J J, Witkamp G J, Comans R N J. Mineral CO2 sequestration by steel slag carbonation[J]. Environmental Science & Technology, 2005, 39(24): 9676-9682. [22] 阎培渝, 郑峰. 温度对补偿收缩复合胶凝材料水化放热特性的影响[J]. 硅酸盐学报, 2006, 34(8): 1006-1010. https://www.cnki.com.cn/Article/CJFDTOTAL-GXYB200608020.htmYan Peiyu, Zheng Feng. Influence of temperature on the hydration heat evolution of shrink-age-compensating complex binders[J]. Journal of the Chinese Ceramic Society, 2006, 34(8): 1006-1010. https://www.cnki.com.cn/Article/CJFDTOTAL-GXYB200608020.htm [23] 杨志杰, 张德, 康栋, 等. 硅钙渣复合地聚物水化机理研究[J]. 矿业科学学报, 2022, 7(5): 577-584. doi: 10.19606/j.cnki.jmst.2022.05.008Yang Zhijie, Zhang De, Kang Dong, et al. Study on hydration mechanism of calcium silicon slag composite geopolymer[J]. Journal of Mining Science and Technology, 2022, 7(5): 577-584. doi: 10.19606/j.cnki.jmst.2022.05.008 -





 下载:
下载: