Optimization of fluorine mine water treatment and fluorine removal mechanism using response surface methodology
-
摘要: 地表水或地下水流经富氟岩石易导致水中氟超标。水中氟超标易导致地方性氟中毒、生态环境的破坏,制约水资源综合利用。基于研制的一种高效除氟药剂,通过响应曲面分析中的Box-Behnken设计除氟试验,优化了pH值、除氟药剂投加量和快速搅拌时间对除氟效果的影响,并通过X射线光电子能谱(XPS)、X射线粉末衍射(XRD)、傅里叶红外光谱(FT-IR)等分析手段对除氟药剂及处理后产生的污泥进行表征分析,探讨了除氟药剂去除氟离子的机理。试验结果表明:最佳除氟条件为pH值6.11,药剂投加量4.15 mL,快速搅拌时间10.90 min,此时可使F-从初始的20 mg/L降至0.453 mg/L,与模型预测的0.445 mg/L基本吻合。除氟机理为,药剂在含氟矿井水中形成铁铝硅氧的多核多羟基络合物,通过氟离子与羟基发生交换、取代羟基,将氟离子固定在多核多羟基络合物中,形成铝硅氧四面体结构的氟化物,实现除氟效果。对比现用的羟基磷灰石吸附为主体的处理工艺,本研究制备的除氟药剂具有处理工艺简单、处理效果稳定、处理成本低等优点。Abstract: Surface water or groundwater flowing through fluorine-rich rocks can easily lead to excessive fluorine in water.Excessive fluoride in water can cause endemic fluorosis, damage of the ecological environment and restrict comprehensive utilization of water resources.In this study, based on a highly effective fluoride removal agent developed by the research group earlier, the box-Behnken method of response surface analysis was applied to design fluoride removal experiments where the influences of pH value, dosage of defluoridation reagent and fast stirring time on fluoride removal effect were optimized.This paper used X-Photoelectron spectroscopy (XPS), X-ray diffraction (XRD) and Fourier transform infrared spectrum (FT-IR) to characterize the defluoridation reagent and the sludge produced after treatment, and discussed the mechanism of fluoride ion removal.The experimental results showed that F-could be reduced from the initial 20 mg/L to 0.453 mg /L under the optimal conditions of fluoride removal (pH value 6.11, dosage 4.15 mL, fast stirring time 10.90 min), which was basically consistent with the predicted value 0.445 mg /L of the model.The defluoridation reagent was mainly through the formation of Fe, Al, Si and O polynuclear polyhydroxyl complex, fluorine-ion exchange with hydroxyl and replace hydroxyl, then fluorine-ion was fixed in the polynuclear polyhydroxyl complex.Meanwhile, fluoride forms a tetrahedral structure of aluminum, silicon and oxygen, and locks fluorine ions in the tetrahedral structure to achieve fluoride removal.Compared with the current main treatment process using hydroxyapatite adsorption, the defluoridation reagent in this study has the advantages of simple treatment process, stable treatment effect and low treatment cost.
-
表 1 实验药剂
Table 1. Experimental reagents
试剂名称 等级 生产厂家 聚合氯化铝 分析纯 德力通净水材料 A 分析纯 国药集团化学试剂有限公司 B 分析纯 德力通净水材料 C 分析纯 福晨化学试剂有限公司 氟化钠 分析纯 上海麦克林生化科技有限公司 盐酸 分析纯 国药集团化学试剂有限公司 氢氧化钠 分析纯 国药集团化学试剂有限公司 聚丙烯酰胺 分析纯 德力通净水材料 表 2 实验仪器
Table 2. Experimental instruments
仪器名称 型号 生产厂家 分析天平 BSA124S 德国赛多利斯集团 恒温鼓风干燥箱 DHG-9243A 上海精其仪器有限公司 恒温磁力搅拌器 B11-1 上海司乐仪器公司 超纯水系统 Direct-Pure RO 赛默飞世尔科技公司 pH计 PHS-3C 上海雷磁仪器公司 浊度计 WZS-186 上海雷磁仪器公司 X射线衍射仪 D8 ADVANCE 布鲁克(北京)科技有限公司 傅立叶红外光谱 Nicolet IS 10 赛默飞世尔科技(中国) 有限公司 X射线光电子能谱 ESCALAB 50Xi 日本Hitachi 氟离子计 PXS-270 上海雷磁仪器公司 六联搅拌机 MY-3000-6M 武汉梅宇仪器公司 表 3 Box-Behnken设计因子及水平
Table 3. Factors and levels of Box-Behnken design
符号 因子 单位 水平 -1 0 +1 a pH值 — 6.0 6.25 6.5 b 药剂投加量 mL 3.5 4.0 4.5 c 快速搅拌时间 min 5 9 13 表 4 BBD实验设计及结果
Table 4. BBD experimental design and results
运行序号 a b/mL c/min 上清液氟离子
浓度/(mg·L-1)1 6.00 3.50 9.00 1.25 2 6.50 3.50 9.00 2.54 3 6.00 4.50 5.00 0.77 4 6.50 4.50 9.00 1.69 5 6.00 4.00 13.00 1.54 6 6.50 4.00 9.00 2.25 7 6.00 4.00 9.00 0.71 8 6.50 4.00 13.00 1.93 9 6.25 3.50 13.00 2.28 10 6.25 4.50 5.00 1.50 11 6.25 3.50 9.00 1.31 12 6.25 4.50 9.00 0.88 13 6.25 4.00 9.00 0.80 14 6.25 4.00 5.00 0.68 15 6.25 4.00 5.00 0.70 16 6.25 4.00 9.00 0.74 17 6.25 4.00 13.00 0.71 表 5 二次响应面回归模型的方差分析
Table 5. Analysis of variance for quadratic response surface regression model
方差来源 平方和 自由度 均方 F值 P值 模型 6.46 9 0.72 112.94 < 0.000 1 a 2.14 1 2.14 336.86 < 0.000 1 b 0.81 1 0.81 126.80 < 0.000 1 c 0.94 1 0.94 147.56 < 0.000 1 ab 0.034 1 0.034 5.38 0.053 4 ac 0.065 1 0.065 10.22 0.015 1 bc 0.031 1 0.031 4.82 0.064 3 a2 0.95 1 0.95 149.84 < 0.000 1 b2 0.55 1 0.55 86.16 < 0.000 1 c2 0.69 1 0.69 108.99 < 0.000 1 残差 0.045 7 0.006 4 失拟项 0.036 3 0.012 5.47 0.067 0 误差 0.008 7 4 0.002 2 总离差 6.51 16 表 6 二次响应面回归模型的可信度分析
Table 6. Credibility analysis of quadratic response surface regression model
参数 数值 相关系数 0.993 2 调整后的相关系数 0.984 4 预测相关系数 0.909 9 信噪比 30.573 标准差 0.080 变异系数/% 6.09 表 7 二次响应面回归模型预测值验证
Table 7. Verification of prediction values of quadratic response surface regression model
除氟药剂
投加量/mLpH值 快速搅拌
时间/min出水氟离子
浓度/(mg·L-1)4.15 6.11 10.9 0.47 4.15 6.11 10.9 0.44 4.15 6.11 10.9 0.45 -
[1] 毛若愚, 郭华明, 贾永锋, 等. 内蒙古河套盆地含氟地下水分布特点及成因[J]. 地学前缘, 2016, 23(2): 260-268. doi: 10.13745/j.esf.2016.02.024Mao Ruoyu, Guo Huaming, Jia Yongfeng, et al. Distribution characteristics and genesis of fluoride groundwater in the Hetao Basin, Inner Mongolia[J]. Earth Science Frontiers, 2016, 23(2): 260-268. doi: 10.13745/j.esf.2016.02.024 [2] 孙亚军, 陈歌, 徐智敏, 等. 我国煤矿区水环境现状及矿井水处理利用研究进展[J]. 煤炭学报, 2020, 45(1): 304-316. doi: 10.13225/j.cnki.jccs.YG19.1654Sun Yajun, Chen Ge, Xu Zhimin, et al. Research progress of water environment, treatment and utilization in coal mining areas of China[J]. Journal of China Coal Society, 2020, 45(1): 304-316. doi: 10.13225/j.cnki.jccs.YG19.1654 [3] 苏双青, 赵焰, 徐志清, 等. 我国煤矿矿井水氟污染现状及除氟技术研究[J]. 能源与环保, 2020, 42(11): 9-14. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZMT202011002.htmSu Shuangqing, Zhao Yan, Xu Zhiqing, et al. Status quo of fluoride pollution of coal mine water in China and research on fluoride removal technology[J]. China Energy and Environmental Protection, 2020, 42(11): 9-14. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZMT202011002.htm [4] 郝春明, 张伟, 何瑞敏, 等. 神东矿区高氟矿井水分布特征及形成机制[J]. 煤炭学报, 2021, 46(6): 1966-1977. doi: 10.13225/j.cnki.jccs.ST21.0160Hao Chunming, Zhang Wei, He Ruimin, et al. Formation mechanisms for elevated fluoride in the mine water in Shendong coal-mining district[J]. Journal of China Coal Society, 2021, 46(6): 1966-1977. doi: 10.13225/j.cnki.jccs.ST21.0160 [5] Khandare D, Mukherjee S. A review of metal oxide nanomaterials for fluoride decontamination from water environment[J]. Materials Today: Proceedings, 2019, 18(3): 1146-1155. [6] Ergun E, Tor A, Cengeloglu Y, et al. Electrodialytic removal of fluoride from water: Effects of process parameters and accompanying anions[J]. Separation and Purification Technology, 2008, 64(2): 147-153. doi: 10.1016/j.seppur.2008.09.009 [7] Ramachandran M, Sabarathinam C, Ulaganthan K, et al. Mapping of fluoride ions in groundwater of Dindigul district, Tamilnadu, India—using GIS technique[J]. Arabian Journal of Geosciences, 2012, 5(3): 433-439. doi: 10.1007/s12517-010-0216-0 [8] Zhang Shengyu, Lyu Ying, Su Xiaosi, et al. Removal of fluoride ion from groundwater by adsorption on lanthanum and aluminum loaded clay adsorbent[J]. Environmental Earth sciences, 2016, 75(5): 401-407. doi: 10.1007/s12665-015-5205-x [9] Hu Yunhu, Xia Chengli, Dong Zhongbing, et al. Geochemical characterization of fluoride in the groundwater of the Huaibei Plain, China[J]. Analytical Letters, 2017, 50(5): 889-903. doi: 10.1080/00032719.2016.1199027 [10] Zhu H J, Wang G Z, Wang H Q, et al. Removal of fluorine from water by using the synthetical iron-aluminum hydroxide complexes[C]//2011 International Conference on Electric Technology and Civil Engineering(ICETCE). Lushan China. IEEE: 6771-6773. [11] 孔垂鹏, 聂玉伦, 胡春. Mg/Al/Fe复合氧化物吸附去除水体中氟化物的研究[J]. 环境工程学报, 2010, 4(1): 110-114. https://www.cnki.com.cn/Article/CJFDTOTAL-HJJZ201001026.htmKong Chuipeng, Nie Yulun, Hu Chun. Enhanced fluoride removal from aqueous solution by mixed oxides derived from Mg/Al/Fe hydrotalcite[J]. Chinese Journal of Environmental Engineering, 2010, 4(1): 110-114. https://www.cnki.com.cn/Article/CJFDTOTAL-HJJZ201001026.htm [12] Islam M, Patel R K. Evaluation of removal efficiency of fluoride from aqueous solution using quick lime[J]. Journal of Hazardous Materials, 2007, 143(1/2): 303-310. [13] 赵焰, 陆梦楠, 孙斌, 等. 含氟矿井水混凝吸附联合除氟技术工业化应用研究[J]. 煤炭科学技术, 2020, 48(9): 166-172. https://www.cnki.com.cn/Article/CJFDTOTAL-MTKJ202009021.htmZhao Yan, Lu Mengnan, Sun Bin, et al. Research on industrial application of coagulation and adsorption combined with fluorine removal technology in fluorine-containing mine water[J]. Coal Science and Technology, 2020, 48(9): 166-172. https://www.cnki.com.cn/Article/CJFDTOTAL-MTKJ202009021.htm [14] Gao W, Majumder M, Alemany L B, et al. Engineered graphite oxide materials for application inwater purification[J]. ACS Applied Materials & Interfaces, 2011, 3(6): 1821-1826. [15] 肖雪峰, 孙永军, 梅凯, 等. 钙沉淀混凝处理太阳能电池生产高氟废水研究[J]. 水处理技术, 2017, 43(5): 30-32. https://www.cnki.com.cn/Article/CJFDTOTAL-SCLJ201705007.htmXiao Xuefeng, Sun Yongjun, Mei Kai, et al. Study on the treatment of solar produced wastewater containing high fluoride by calcium coagulation-sedimentation[J]. Technology of water treatment, 2017, 43(5): 30-32. https://www.cnki.com.cn/Article/CJFDTOTAL-SCLJ201705007.htm [16] 章丽萍, 吴胜念, 宋学京, 等. 难沉降钨矿选矿废水处理研究[J]. 矿业科学学报, 2020, 5(6);688-696. doi: 10.19606/j.cnki.jmst.2020.06.012Zhang Liping, Wu Shengnian, Song Xuejing, et al. Study on treatment of difficult settling wastewater from tungsten mineral processing[J]. Journal of Mining Science and Technology, 2020, 5(6): 688-696 doi: 10.19606/j.cnki.jmst.2020.06.012 [17] 蒋斌斌, 高昊, 杜坤, 等. 煤基复合絮凝剂对高浊度高矿化度矿井水的絮凝效果研究[J]. 矿业科学学报, 2020, 5(6): 682-687. doi: 10.19606/j.cnki.jmst.2020.06.011Jiang Binbin, Gao Hao, Du Kun, et al. Research on flocculation effect of fly ash composite on high turbidity and high salinity mine water[J]. Journal of Mining Science and Technology, 2020, 5(6): 682-687 doi: 10.19606/j.cnki.jmst.2020.06.011 [18] Koilraj P, Kannan S. Aqueous fluride removal using Zn Cr layered double hydroxides and their polymeric composites: Batch and column studies[J]. Chemical Engineering Journal, 2013, (234): 406-415. [19] 刘敏. 羟基磷灰石去除煤矿矿井水氟化物工艺研究及参数优化[J]. 煤化工, 2021, 49(1): 80-85. https://www.cnki.com.cn/Article/CJFDTOTAL-MHGZ202101025.htmLiu Min. Study on process and parameter optimization of defluorination from coal mine water with hydroxyapatite[J]. Coal Chemical industry, 2021, 49(1): 80-85. https://www.cnki.com.cn/Article/CJFDTOTAL-MHGZ202101025.htm [20] 刘锐平. 饮用水氟污染控制原理与技术[J]. 应用生态学报, 2019, 30(1): 30-36. https://www.cnki.com.cn/Article/CJFDTOTAL-YYSB201901005.htmLiu Ruiping. Principle and techniques for fluoride pollution control in drinking water[J]. Chinese Journal of Applied Ecology, 2019, 30(1): 30-36. https://www.cnki.com.cn/Article/CJFDTOTAL-YYSB201901005.htm [21] Sehn P. Fluoride removal with extra low energy reverse osmosis membranes: three years of large scale field experience in Finland[J]. Desalination, 2008, 223(1/2/3): 73-84. [22] Huang C J, Liu J C. Precipitate flotation of fluoride-containing wastewater from a semiconductor manufacturer[J]. Water Research, 1999, 33(16): 3403-3412. [23] 郑搏英, 张襄, 朱希坤, 等. 一种深度除氟剂及其制备方法与应用: CN111573806A[P]. 2020-08-25. [24] 王恒, 杨友鑫, 康勇锋, 等. 一种高效除氟复配药剂: CN109574177A[P]. 20190405. [25] 章丽萍, 吴二勇, 姚瑞涵, 等. 高效除氟药剂对神东矿区含氟矿井水的处理研究[J]. 干旱区资源与环境, 2022, 36(2): 84-90. https://www.cnki.com.cn/Article/CJFDTOTAL-GHZH202202012.htmZhang Liping, Wu Eryong, Yao Ruihan, et al. Treatment of fluoride-containing mine water from Shendong coalmine with high efficiency defluoridation agent[J]. Journal of Arid Land Resources and Environment, 2022, 36(2): 84-90. https://www.cnki.com.cn/Article/CJFDTOTAL-GHZH202202012.htm [26] 彭赵旭, 牛宁琪, 王炬, 等. 响应面法优化石灰处理高氟废水的研究[J]. 河南师范大学学报: 自然科学版, 2022, 50(1): 108-114. https://www.cnki.com.cn/Article/CJFDTOTAL-HNSX202201012.htmPeng Zhaoxu, Niu Ningqi, Wang Ju, et al. Study on optimizing lime treatment of high fluoride concentration wastewater by response surface methodology[J]. Journal of Henan Normal University: Natural Science Edition, 2022, 50(1): 108-114. https://www.cnki.com.cn/Article/CJFDTOTAL-HNSX202201012.htm [27] Khobragade M U, Nayak A K, Pal A. Application of response surface methodology to evaluate the removal efficiency of Mn(Ⅱ), Ni(Ⅱ), and Cu(Ⅱ) by surfactant-modified alumina[J]. Clean Technologies and Environmental Policy, 2016, 18(4): 1003-1020. [28] 严博文, 叶长文, 龚锐, 等. 响应曲面分析优化改性粉煤灰漂珠对水中氟的吸附性能及机理研究[J]. 环境科学研究, 2019, 32(4): 709-717. https://www.cnki.com.cn/Article/CJFDTOTAL-HJKX201904021.htmYan Bowen, Ye Changwen, Gong Rui, et al. Optimization study of adsorption parameters for removal of fluoride by fly ash cenospheres modified with calcium using response surface methodology[J]. Research of Environmental Sciences, 2019, 32(4): 709-717. https://www.cnki.com.cn/Article/CJFDTOTAL-HJKX201904021.htm [29] 由昆, 范维利, 傅金祥, 等. 载锰活性氧化铝除氟降铁效能研究[J]. 中国给水排水, 2020, 36(23): 42-51. https://www.cnki.com.cn/Article/CJFDTOTAL-GSPS202023012.htmYou Kun, Fan Weili, Fu Jinxiang, et al. Fluorine and iron removal efficiency of manganese-loaded activated alumina[J]. China Water & Wastewater, 2020, 36(23): 42-51. https://www.cnki.com.cn/Article/CJFDTOTAL-GSPS202023012.htm [30] Jiang Shuihua, Li Dianqing, Zhou Chuangbing, et al. Capabilities of stochastic response surface method and response surface method in reliability analysis[J]. Structural Engineering and Mechanics, 2014, 49(1): 111-128. [31] Zhang Liping, Jiao Xifei, Wu Shengnian, et al. Study on optimization of Tungsten Ore flotation wastewater treatment by response surface method (RSM)[J]. Minerals, 2021, 11(2): 184-198. [32] 王文静, 仲丽娟, 黄保平, 等. 地表水强化混凝除氟方案[J]. 净水技术, 2020, 39(3): 95-98, 151. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSJS202003022.htmWang Wenjing, Zhong Lijuan, Huang Baoping, et al. Solutions of fluoride removal of surface water by enhanced coagulation process[J]. Water Purification Technology, 2020, 39(3): 95-98, 151. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSJS202003022.htm [33] 郑洁. 新型除氟聚合铝基吸附剂特性及机理研究[D]. 呼和浩特: 内蒙古大学, 2020. [34] 张建成, 刘利波. 混凝剂在选煤厂煤泥水处理中的应用及机理研究[J]. 中国煤炭, 2018, 44(1): 89-93. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGME201801020.htmZhang Jiancheng, Liu Libo. Mechanism study and application on coagulant of coal slurry treatment in coal preparation plant[J]. China Coal, 2018, 44(1): 89-93. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGME201801020.htm [35] 张海阳, 高柏, 樊骅, 等. XRD和FTIR对Ce/γ-Al2O3除氟除砷的机理研究[J]. 光谱学与光谱分析, 2020, 40(9): 2869-2874. https://www.cnki.com.cn/Article/CJFDTOTAL-GUAN202009040.htmZhang Haiyang, Gao Bai, Fan Hua, et al. Mechanism of fluoride and arsenic removal by Ce/γ-Al2O3 based on XRD and FTIR[J]. Spectroscopy and Spectral Analysis, 2020, 40(9): 2869-2874. https://www.cnki.com.cn/Article/CJFDTOTAL-GUAN202009040.htm [36] 郜玉楠, 茹雅芳, 王静, 等. 微米氧化锆/沸石分子筛处理高氟地下水的研究[J]. 中国给水排水, 2020, 36(3): 49-53. https://www.cnki.com.cn/Article/CJFDTOTAL-GSPS202003012.htmGao Yunan, Ru Yafang, Wang Jing, et al. Treatment of high fluoride groundwater by micron zirconia/zeolite molecular sieve[J]. China Water & Wastewater, 2020, 36(3): 49-53. https://www.cnki.com.cn/Article/CJFDTOTAL-GSPS202003012.htm [37] Aoudj S, Drouiche N, Hecini M, et al. Coagulation as a post-treatment method for the defluoridation of photovoltaic cell manufacturing wastewater[J]. Procedia Engineering, 2012, 33: 111-120. [38] 张怡晓. 煤矿矿井废水絮凝药剂的优化研究[D]. 北京: 北京林业大学, 2019. -
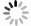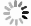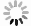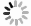





 下载:
下载: