On stability and synergistic effect of nano-bubbles in micro-silica flotation
-
摘要: 针对微硅粉的提纯问题,提出了在微硅粉浮选中引入纳米气泡以提升浮选回收率的方案,分析了纳米气泡对浮选提纯微硅粉的影响;使用纳米颗粒跟踪分析(NTA)测定纳米气泡的稳定性和油酸钠对其稳定性的影响。结果表明:在捕收剂等量的情况下,在微硅粉浮选中引入纳米气泡,可以提高纳米SiO2的回收率,在捕收剂用量为1.5 g/L时,纳米SiO2回收率达到最大,为70.2 %;油酸钠可以提高水中纳米气泡的稳定性和分散性;纳米气泡可以提高浮选回收率,可能是由于纳米气泡与浮选中产生的普通浮选气泡发生协同作用。Abstract: In order to solve the purification problem of microsilica powder and improve the flotation recovery, this study introduced nano-bubbles into the flotation of micro-silicon powder, and analyzed the effect of nano-bubbles on flotation purification of microsilica powder.Nanoparticle Tracking Analysis (NTA) was used to determine the stability of nano-bubbles and the effect of sodium oleate on their stability.Results show that with the same amount of collector, the recovery of SiO2 can be improved by introducing nano-bubbles into the flotation of micro-silica powder; When the amount of collector was 1.5 g/L, the recovery of SiO2 reached the maximum of 70.2 %; Sodium oleate can improve the stability and dispersion of nano-bubbles in water; Nanobubbles can improve flotation recovery since there is a synergistic effect between nano bubbles and ordinary flotation bubbles produced in flotation.
-
Key words:
- microsilica /
- fume flotation /
- purification /
- nanobubbles /
- nanoparticle tracking analysis
-
表 1 微硅粉的化学成分组成
Table 1. The chemical composition of microsilica
成分 SiO2 Al2O3 Fe2O3 MgO CaO K2O Na2O C 质量分数/% 85.89 0.27 5.23 0.31 0.4 0.47 0.15 4.11 表 2 各组实验条件
Table 2. Test conditions of each group
组别 水 药剂 放置时间/min 1 纳米气泡水 无 30 2 纳米气泡水 无 90 3 纳米气泡水 无 180 4 纳米气泡水 油酸钠 30 5 纳米气泡水 油酸钠 90 6 纳米气泡水 油酸钠 180 7 蒸馏水 无 — 表 3 各组样品纳米气泡稳定性表征
Table 3. Characterization of the stability of nanobubbles under different conditions
类别 样本编号 1 2 3 4 5 6 7 粒径平均数/nm 153.3 167.6 229.7 127.5 140.7 145.1 107.9 粒径众数/nm 147.0 121.6 147.2 86.2 86.5 92.9 71.1 粒径标准差/nm 42.7 63.6 100.7 71.0 82.5 71.3 56.0 d10/nm 100.9 99.0 123.4 81.0 83.3 85.3 68.6 d50/nm 150.5 153.5 210.9 103.4 111.1 123.2 91.1 d90/nm 201.8 258.2 380.0 194.2 217.0 242.3 153.9 总气泡浓度/ (108·mL-1) 2.79 2.55 1.24 6.98 3.86 2.53 0.26 -
[1] 王杰, 魏奎先, 马文会, 等. 工业微硅粉应用及提纯研究进展[J]. 材料导报, 2020, 34(23): 23081-23087. doi: 10.11896/cldb.19090058Wang Jie, Wei Kuixian, Ma Wenhui, et al. Research progress in purification and application of industrial silica fume[J]. Materials Reports, 2020, 34(23): 23081-23087. doi: 10.11896/cldb.19090058 [2] 刘丽娟, 李铁龙, 金朝晖. 硅微粉的提纯实验研究[J]. 环境工程学报, 2010, 4(12): 2879-2882. https://www.cnki.com.cn/Article/CJFDTOTAL-HJJZ201012049.htmLiu Lijuan, Li Tielong, Jin Zhaohui. Experimental study on purifying silica fume[J]. Chinese Journal of Environmental Engineering, 2010, 4(12): 2879-2882. https://www.cnki.com.cn/Article/CJFDTOTAL-HJJZ201012049.htm [3] 刘晓华, 盖国胜. 微硅粉在国内外应用概述[J]. 铁合金, 2007, 38(5): 41-44. doi: 10.3969/j.issn.1001-1943.2007.05.011Liu Xiaohua, Gai Guosheng. Application of micro-silica at home and abroad[J]. Ferro-Alloys, 2007, 38(5): 41-44. doi: 10.3969/j.issn.1001-1943.2007.05.011 [4] 张金梁, 卢萍, 杨桂生, 等. 微硅粉性能表征与综合利用研究现状分析[J]. 矿冶, 2020, 29(4): 116-122. https://www.cnki.com.cn/Article/CJFDTOTAL-KYZZ202004023.htmZhang Jinliang, Lu Ping, Yang Guisheng, et al. Properties characterization and comprehensive utilization status analysis of silica fume[J]. Mining and Metallurgy, 2020, 29(4): 116-122. https://www.cnki.com.cn/Article/CJFDTOTAL-KYZZ202004023.htm [5] Kalantari B, Prasad A, Huat B B K. Cement and silica fume treated columns to improve peat ground[J]. Arabian Journal for Science and Engineering, 2013, 38(4): 805-816. doi: 10.1007/s13369-012-0369-0 [6] 王小楠. 微硅粉的资源化利用及其吸附性能的研究[D]. 青岛: 山东科技大学, 2011. [7] 唐建新, 吕艳红, 李传山, 等. 固体废弃物微硅粉应用研究进展[J]. 现代技术陶瓷, 2013, 34(4): 17-21. doi: 10.3969/j.issn.1005-1198.2013.04.004Tang Jianxin, Lü Yanhong, Li Chuanshan, et al. Research progress in application of solid waste silicon ash[J]. Advanced Ceramics, 2013, 34(4): 17-21. doi: 10.3969/j.issn.1005-1198.2013.04.004 [8] 张启万. 硅微粉的生产与应用[J]. 铁合金, 2005, 36(3): 14-17. doi: 10.3969/j.issn.1001-1943.2005.03.004Zhang Qiwan. Produce and application of silicon tiny powder[J]. Ferro-alloys, 2005, 36(3): 14-17. doi: 10.3969/j.issn.1001-1943.2005.03.004 [9] 铁生年, 姜子炎, 汪长安. 矿热炉冶炼铁合金回收微硅粉粉体应用研究与发展现状[J]. 硅酸盐通报, 2012, 31(6): 1491-1497. https://www.cnki.com.cn/Article/CJFDTOTAL-GSYT201206032.htmTie Shengnian, Jiang Ziyan, Wang Changan. The application research and present development situation of silica fume powder produced from submerged arc furnace smelting ferroalloy[J]. Bulletin of the Chinese Ceramic Society, 2012, 31(6): 1491-1497. https://www.cnki.com.cn/Article/CJFDTOTAL-GSYT201206032.htm [10] 高建平, 张启轩, 冯超. 冶金固体废弃物二氧化硅微粉资源化利用: 硅系铁合金电炉烟气净化及回收微硅粉的利用[J]. 冶金标准化与质量, 2007, 45(2): 5-7, 15. https://www.cnki.com.cn/Article/CJFDTOTAL-YJBL200702001.htmGao Jianping, Zhang Qixuan, Feng Chao. The utilization of metallurgical solid waste SiO2 fine particle[J]. Metallurgical Standardization & Quality, 2007, 45(2): 5-7, 15. https://www.cnki.com.cn/Article/CJFDTOTAL-YJBL200702001.htm [11] 马艳芳, 李宁, 常钧. 硅灰性能及其再利用的研究进展[J]. 无机盐工业, 2009, 41(10): 8-10. https://www.cnki.com.cn/Article/CJFDTOTAL-WJYG200910002.htmMa Yanfang, Li Ning, Chang Jun. Research progress on properties and recycling of silica fume[J]. Inorganic Chemicals Industry, 2009, 41(10): 8-10. https://www.cnki.com.cn/Article/CJFDTOTAL-WJYG200910002.htm [12] 孙宁, 李俊翰, 杨绍利, 等. 工业微硅粉的提纯与应用技术研究进展[J]. 无机盐工业, 2017, 49(8): 5-9. https://www.cnki.com.cn/Article/CJFDTOTAL-WJYG201708002.htmSun Ning, Li Junhan, Yang Shaoli, et al. Research progress in purification and application technology of industrial silica fume[J]. Inorganic Chemicals Industry, 2017, 49(8): 5-9. https://www.cnki.com.cn/Article/CJFDTOTAL-WJYG201708002.htm [13] 冯柳毅, 林荣毅, 田登超, 等. 酸浸工艺脱除微硅粉杂质离子及其对热碱溶解过程的强化[J]. 化工进展, 2015, 34(12): 4379-4384, 4390.Feng Liuyi, Lin Rongyi, Tian Dengchao, et al. Impurity ions of micro silicon powder removal by acid leaching and intensification to the hot alkali dissolving process[J]. Chemical Industry and Engineering Progress, 2015, 34(12): 4379-4384, 4390. [14] 周利民. 水中纳米气泡的气体状态与稳定机理的探究[D]. 上海: 中国科学院大学(中国科学院上海应用物理研究所), 2019. [15] Craig V S J. Very small bubbles at surfaces—the nanobubble puzzle[J]. Soft Matter, 2011, 7(1): 40-48. [16] Alheshibri M, Qian J, Jehannin M, et al. A history of nanobubbles[J]. Langmuir, 2016, 32(43): 11086-11100. [17] 曾维能, 任浏祎, 魏鹏刚, 等. 微纳米气泡对典型细粒氧化矿物浮选的影响及机理[J]. 金属矿山, 2020(10): 156-160. https://www.cnki.com.cn/Article/CJFDTOTAL-JSKS202010021.htmZeng Weineng, Ren Liuyi, Wei Penggang, et al. Effects and mechanism of micro-nano bubbles on typical fine oxidized minerals flotation[J]. Metal Mine, 2020(10): 156-160. https://www.cnki.com.cn/Article/CJFDTOTAL-JSKS202010021.htm [18] Knüpfer P, Ditscherlein L, Peuker U A. Nanobubble enhanced agglomeration of hydrophobic powders[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2017, 530: 117-123. [19] Xiao Wei, Wang Xingxing, Zhou Limin, et al. Influence of mixing and nanosolids on the formation of nanobubbles[J]. Journal of Physical Chemistry B, 2019(1): 317-323. [20] Sobhy A, Tao D. Nanobubble column flotation of fine coal particles and associated fundamentals[J]. International Journal of Mineral Processing, 2013(11): 109-116. [21] Rahman A, Ahmad K D, Mahmoud A, et al. Nano-micro-bubble flotation of fine and ultrafine chalcopyrite particles[J]. International Journal of Mining Science and Technology, 2014(4): 559-566. [22] Calgaroto S, Azevedo A, Rubio J. Flotation of quartz particles assisted by nanobubbles[J]. International Journal of Mineral Processing, 2015, 137: 64-70. [23] Nirmalkar N, Pacek A W, Barigou M. Interpreting the interfacial and colloidal stability of bulk nanobubbles[J]. Soft Matter, 2018, 14(47): 9643-9656. [24] Calgaroto S, Wilberg K Q, Rubio J. On the nanobubbles interfacial properties and future applications in flotation[J]. Minerals Engineering, 2014, 60: 33-40. [25] 李硕夫. 浮选过程中颗粒与气泡在不同流态下碰撞及粘附特性研究[D]. 北京: 北京科技大学, 2021. -
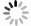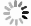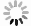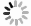





 下载:
下载: