Investigating the performance of dry chemically modified steel slag powder and pastes by formic acid
-
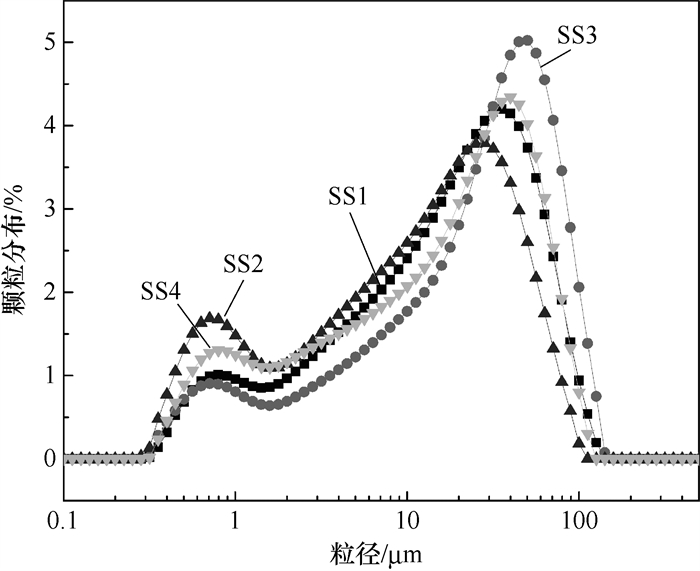
摘要: 探明化学改性钢渣粉的活性和力学性能对提升钢渣利用具有重要意义。研究选取4种钢渣粉采用4 % 的甲酸溶液进行干法化学改性,采用背散射电子显微镜、水化量热仪、X射线衍射仪、傅里叶变换红外光谱仪表征了改性前后钢渣粉及其浆体性能的变化。结果发现,不同钢渣粉矿物组成差异较大,制备的浆体3 d和7 d抗压强度均较低,72 h的水化热在10~40 J/g间波动;但经过甲酸改性后,其3 d抗压强度提升率都超过200 %,72 h水化热均提升至50 J/g以上,提升率均超过80 %,钢渣粉中的Ca(OH)2均能参与反应并生成甲酸钙,而其中的活性硅酸盐未受到显著影响。Abstract: Understanding the composition and reactivity of formic acid modified steel slag powder (SS) from micro scale is critical to extending the utilization of steel slag. In this study, four representative sources of SS were selected, which were dry chemically modified by formic acid (FA) with a mass fraction of 4 % on the basis of SS. The backscatter electron microscope, hydration calorimeter, X-ray diffractometer and fourier transform infrared spectrometer were applied to characterize the SS and SS pastes before and after modification. Results show that the 3 d and 7 d compressive strength of the different sources of SS pastes are low, and the 72 h cumulative hydration heat varies between 10~40 J/g due to their different mineral compositions. However, after FC modification, the 3 d compressive strength increases by more than 200 %, and the 72 h cumulative hydration heat rises to above 50 J/g with an increase above 80 %.it is found that the Ca(OH)2 in SS can react with FA to produce calcium formate, while the calcium silicates in SS are not significantly affected.
-
Key words:
- steel slag powder /
- chemical modification /
- reactivity /
- mechanism
-
表 1 钢渣粉的化学组成
Table 1. Chemical composition of SS
% 钢渣 CaO SiO2 Al2O3 Fe2O3 SO3 MgO Na2O K2O TiO2 P 总量 SS1 43.49 17.65 8.32 21.39 1.46 5.92 0.14 0.07 0.87 0.67 99.98 SS2 49.58 13.41 2.39 23.72 0.28 7.94 0.08 0.03 1.44 1.12 99.99 SS3 46.13 13.90 1.86 29.60 0.30 5.77 0.28 0.08 0.95 1.06 99.93 SS4 47.39 13.09 1.35 29.04 0.65 6.05 0.35 0.07 0.88 1.00 99.87 表 2 改性前后钢渣浆体的活性和抗压强度对比
Table 2. Comparation the activity and compressive strength of SS pastes before and after modification
试样 72 h水化热/(J·g-1) 抗压强度/MPa 3 d 28 d SS1 36.29 0.5 8.4 SS2 13.99 0.4 5.0 SS3 10.73 0.3 4.5 SS4 20.45 0.4 6.5 SS1M 68.91 1.9 8.0 SS2M 58.19 1.3 4.5 SS3M 51.86 0.9 3.8 SS4M 53.57 1.5 7.5 -
[1] 王栋民. 固废与生态材料的未来发展(代序)[J]. 硅酸盐通报, 2020, 39(8): 2357-2358. https://www.cnki.com.cn/Article/CJFDTOTAL-GSYT202008001.htmWang Dongmin. Future development of solid waste and ecological materials (preface)[J]. Bulletin of the Chinese Ceramic Society, 2020, 39(8): 2357-2358. https://www.cnki.com.cn/Article/CJFDTOTAL-GSYT202008001.htm [2] Wei X, Qiu R, Liang Y, et al. Roadmap to carbon emissions neutral industrial parks: energy, economic and environmental analysis[J]. Energy, 2022, 238: 121732. doi: 10.1016/j.energy.2021.121732 [3] 叶家元, 张文生, 史迪, 等. 钢渣碳化砖的碱激发-碳化协同效应影响因素[J]. 硅酸盐学报, 2019, 47(11): 1582-1592. https://www.cnki.com.cn/Article/CJFDTOTAL-GXYB201911009.htmYe Jiayuan, Zhang Wensheng, Shi Di, et al. Synergistic effect of alkali-activation and carbonation on carbonated steel slag bricks[J]. Journal of the Chinese Ceramic Society, 2019, 47(11): 1582-1592. https://www.cnki.com.cn/Article/CJFDTOTAL-GXYB201911009.htm [4] Jiang Y, Ling T C, Shi C, et al. Characteristics of steel slags and their use in cement and concrete-a review[J]. Resources, Conservation and Recycling, 2018, 136: 187-197. doi: 10.1016/j.resconrec.2018.04.023 [5] Zhao J H, Yan P Y, Wang D M. Research on mineral characteristics of converter steel slag and its comprehensive utilization of internal and external recycle[J]. Journal of Cleaner Production, 2017, 156: 50-61. doi: 10.1016/j.jclepro.2017.04.029 [6] Li B L, Wang Y H, Yang L, et al. Sulfate resistance and hydration products of steam cured steel slag blended cement mortar under dry-wet cycle[J]. Journal of Sustainable Cement-Based Materials, 2019, 8(6): 353-366. doi: 10.1080/21650373.2018.1564709 [7] Li J X, Yu Q J, Wei J X, et al. Structural characteristics and hydration kinetics of modified steel slag[J]. Cement and Concrete Research, 2011, 41(3): 324-329. doi: 10.1016/j.cemconres.2010.11.018 [8] Sun J W, Zhang Z Q, Zhuang S Y, et al. Hydration properties and microstructure characteristics of alkali-activated steel slag[J]. Construction and Building Materials, 2020, 241: 118141. doi: 10.1016/j.conbuildmat.2020.118141 [9] Humbert P S, Castro-Gomes J. CO2 activated steel slag-based materials: a review[J]. Journal of Cleaner Production, 2019, 208: 448-457. doi: 10.1016/j.jclepro.2018.10.058 [10] Tsakiridis P E, Papadimitriou G D, Tsivilis S, et al. Utilization of steel slag for Portland cement clinker production[J]. Journal of Hazardous Materials, 2008, 152(2): 805-811. doi: 10.1016/j.jhazmat.2007.07.093 [11] Huo B B, Li B L, Chen C, et al. Surface etching and early age hydration mechanisms of steel slag powder with formic acid[J]. Construction and Building Materials, 2021, 280: 122500. doi: 10.1016/j.conbuildmat.2021.122500 [12] Huo B B, Li B L, Chen C, et al. Morphological and mineralogical insights into acetic acid modifying and hydraulic process on steel slag for enhanced reactivity[J]. Construction and Building Materials, 2021, 307: 125004. doi: 10.1016/j.conbuildmat.2021.125004 [13] Jiang L, Bao Y W, Chen Y H, et al. Structural characteristics and hydration kinetics of oxidized steel slag in a CaO-FeO-SiO2-MgO system[J]. High Temperature Materials and Processes, 2019, 38(2019): 290-300. doi: 10.1515/htmp-2017-0163 [14] Tossavainen M, Engstrom F, Yang Q, et al. Characteristics of steel slag under different cooling conditions[J]. Waste Management, 2007, 27(10): 1335-1344. doi: 10.1016/j.wasman.2006.08.002 [15] Martins A C P, Franco de Carvalho J M, Costa L C B, et al. Steel slags in cement-based composites: an ultimate review on characterization, applications and performance[J]. Construction and Building Materials, 2021, 291: 123265. doi: 10.1016/j.conbuildmat.2021.123265 [16] Zhuang S Y, Wang Q. Inhibition mechanisms of steel slag on the early-age hydration of cement[J]. Cement and Concrete Research, 2021, 140: 106283. doi: 10.1016/j.cemconres.2020.106283 [17] Huo B B, Luo Y L, Li B L, et al. Influence of particle size on the reactivity of chemical modified steel slag powder[J]. Journal of Sustainable Cement-Based Materials, 2022: 1-9. [18] Cao L, Shen W G, Huang J Q, et al. Process to utilize crushed steel slag in cement industry directly: multi-phased clinker sintering technology[J]. Journal of Cleaner Production, 2019, 217: 520-529. doi: 10.1016/j.jclepro.2019.01.260 [19] 侯新凯, 袁静舒, 杨洪艺, 等. 钢渣中水化惰性矿物的化学物相分析[J]. 硅酸盐学报, 2016, 44(5): 651-657. https://www.cnki.com.cn/Article/CJFDTOTAL-GXYB201605006.htmHou Xinkai, Yuan Jingshu, Yang Hongyi, et al. Chemical phase analysis of nonhydraulic minerals in steel slag[J]. Journal of the Chinese Ceramic Society, 2016, 44(5): 651-657. https://www.cnki.com.cn/Article/CJFDTOTAL-GXYB201605006.htm [20] 刘泽, 李丽, 邵宁宁, 等. 钢渣-粉煤灰水热合成方沸石及其性能表征[J]. 矿业科学学报, 2018, 3(5): 508-514. http://kykxxb.cumtb.edu.cn/article/id/178Liu Ze, Li Li, Shao Ningning, et al. Hydrothermal synthesis of analcime from steel slag-fly ash and its performance characterization[J]. Journal of Mining Science and Technology, 2018, 3(5): 508-514. http://kykxxb.cumtb.edu.cn/article/id/178 -





 下载:
下载: