Preparation of coffee residual based biochar and its removal of fluoride ions from mine water
-
摘要: 近年来矿井水中氟离子含量超标问题尤为突出,不仅影响矿井水处理后的回用水质,而且危害人体健康。为了同时实现固废资源化利用和矿井水中氟离子的去除,本文以废弃咖啡渣为原料,采用化学活化法制备了一种新型矿井水中氟离子的吸附剂——咖啡渣生物质炭(CRB),通过单因素法优化了制备过程中的活化剂种类、活化温度和活化浓度;通过动力学和等温吸附实验对比分析了优化前后CRB对氟离子的吸附效能;通过表面物化特性表征探究了优化后的CRB(O-CRB)对氟离子的吸附机理。结果表明:当采用氯化锌为活化剂、浓度为4 mol/L、活化温度为400 ℃时,获得了吸附性能最优的O-CRB,其对氟离子的最大吸附量为2.20 mg/g,是活化前CRB的1.30倍;与优化前相比,O-CRB的中孔孔容量与比表面积明显增加,含氧官能基团(-COOH)含量和表面Zeta电位升高,且吸附氟离子后O-CRB表面的氯离子含量明显降低(3.27 % 至0.61 %)、氟离子含量明显增加(0.76 % 至10.34 %),从而可推测出O-CRB吸附氟离子的主要机理为静电吸附和离子交换。Abstract: In recent years, the problem of excessive fluoride in mine water is particularly prominent, which not only affects the water quality of reuse after treatment, but also endangers human health.Therefore, the study herein developed a novel adsorbent for fluoride -coffee residual based biochar(CRB)by applying the waste coffee residue as raw material and chemical activation, to realize the purpose of solid waste resource utilization and fluoride removal from mine water at the same time.The authors firstly optimized the preparation protocols of CRB through single factor, including the type of activator, activator concentration and activation temperature; then evaluated the adsorption efficiency of fluoride for CRB before and after optimization by kinetic and isothermal adsorption experiments; finally investigated the adsorption mechanism of fluoride for CRB after optimization(O-CRB)by surface physicochemical characterization.The results showed that the more favorable CRB for fluoride was prepared at the ZnCl2 concentration of 4 mol/L and the activation temperature of 400 ℃.The maximum adsorption capacity of O-CRB for fluoride was 2.20 mg/g, which was 1.30 times higher than that of CRB before activation.In addition, compared with CRB, O-CRB exhibited higher amount of mesopore volume and specific surface area, as well as more oxygen containing functional groups(-COOH)and surface Zeta potential.Thus, the amount of chloride on the O-CRB surface decreased obviously(3.27 % to 0.61 %)after fluoride adsorption, whereas the amount of fluoride increased significantly(0.76 % to 10.34 %).This suggested that the adsorption of fluoride for O-CRB was mainly attributed to the electrostatic adsorption and ion exchange.
-
Key words:
- coffee residuals /
- biochar /
- mine water /
- fluoride /
- adsorption mechanism
-
表 1 CRB与O-CRB的动力学吸附和等温吸附模型拟合参数
Table 1. Fitting parameters of kinetic and isothermal adsorption model for CRB and O-CRB
类型 准一级动力学 准二级动力学 Langmuir Freundlich K1/min-1 R2 K2/g(mg min)-1 R2 qm/(mg·g-1) KL/(L·mg-1) R2 1/n KF R2 CRB 0.003 41 0.775 0.078 4 0.999 1.69 3.18 0.999 0.200 1.11 0.952 O-CRB 0.003 63 0.815 0.049 2 0.999 2.20 7.50 0.999 0.180 1.61 0.860 表 2 3种生物质炭的比表面积、孔容孔径分布和表面元素分析
Table 2. Specific surface area, pore volume distribution and surface element analysis of three biochars
表面特性 CRB O-CRB F-OCRB 微孔孔容/(cm3 ·g-1) 0.000 15 0.000 069 0.000 26 中孔孔容/(cm3· g-1) 0.002 5 0.007 6 0.005 3 总孔容/(cm3· g-1) 0.002 6 0.007 7 0.005 6 比表面积/(m2· g-1) 0.17 2.49 1.89 平均孔径/ nm 60.44 12.31 11.83 cC/% 83.05 83.47 75.77 cN/% 3.41 2.43 1.68 cO/% 12.61 10.11 11.59 cCl/% 0.17 3.27 0.61 cF/% 0.76 0.73 10.34 -
[1] Prabhu S M, Koilraj P, Sasaki K. Synthesis of sucrose-derived porous carbon-doped ZrxLa1-xOOH materials and their superior performance for the simultaneous immobilization of arsenite and fluoride from binary systems[J]. Chemical Engineering Journal, 2017, 325: 1-13. doi: 10.1016/j.cej.2017.05.052 [2] 苏双青, 赵焰, 徐志清, 等. 我国煤矿矿井水氟污染现状及除氟技术研究[J]. 能源与环保, 2020, 42(11): 5-10. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZMT202011002.htmSu Shuangqing, Zhao Yan, Xu Zhiqing, et al. Status quo of fluoride pollution of coal mine water in China and research on fluoride removal technology[J]. China Energy and Environmental Protection, 2020, 42(11): 5-10. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZMT202011002.htm [3] 赵焰, 陆梦楠, 孙斌, 等. 含氟矿井水混凝吸附联合除氟技术工业化应用研究[J]. 煤炭科学技术, 2020, 48(9): 166-172. https://www.cnki.com.cn/Article/CJFDTOTAL-MTKJ202009021.htmZhao Yan, Lu Mengnan, Sun Bin, et al. Research on industrial application of coagulation and adsorption combined with fluorine removal technology in fluorine-containing mine water[J]. Coal Science and Technology, 2020, 48(9): 166-172 https://www.cnki.com.cn/Article/CJFDTOTAL-MTKJ202009021.htm [4] 解国强. 对布尔台煤田地质勘查中水文地质问题的研究[J]. 内蒙古煤炭经济, 2015(2): 208-210. doi: 10.3969/j.issn.1008-0155.2015.02.128Xie Guoqiang. Study on hydrogeological problems in the geological exploration of Bultai Coalfield[J]. Inner Mongolia Coal Economy, 2015 (2): 208-210. doi: 10.3969/j.issn.1008-0155.2015.02.128 [5] 叶群峰, 周小玲. 金华市萤石矿区土壤氟污染评价[J]. 环境科学, 2015, 36(7): 2648-2654. https://www.cnki.com.cn/Article/CJFDTOTAL-HJKZ201507049.htmYe Qunfeng, Zhou Xiaoling. Assessment of soil fluorine pollution in Jinhua fluorite ore areas[J]. Environmental Science, 2015, 36(7): 2648-2654. https://www.cnki.com.cn/Article/CJFDTOTAL-HJKZ201507049.htm [6] Iriel A, Bruneel S P, Schenone N, et al. The removal of fluoride from aqueous solution by a lateritic soil adsorption: Kinetic and equilibrium studies[J]. Ecotoxicology and Environmental Safety, 2018, 149: 166-172. doi: 10.1016/j.ecoenv.2017.11.016 [7] Sehn P. Fluoride removal with extra low energy reverse osmosis membranes: three years of large scale field experience in Finland[J]. Desalination, 2008, 223(1/2/3): 73-84. http://www.onacademic.com/detail/journal_1000034070714710_6d04.html [8] Bhadja V, Trivedi J S, Chatterjee U. Efficacy of polyethylene Interpolymer membranes for fluoride and arsenic ion removal during desalination of water via electrodialysis[J]. RSC Advances, 2016, 6(71): 67118-67126. doi: 10.1039/C6RA11450D [9] 程浩铭, 张翠玲, 任昊晔, 等. 化学沉淀法处理高氟废水的工艺条件优化[J]. 兰州交通大学学报, 2018, 37(5): 80-84. doi: 10.3969/j.issn.1001-4373.2018.05.014Cheng Haoming, Zhang Cuiling, Ren Haoye, et al. Optimization of the technological conditions for the treatment of high fluoride wastewater by the chemical precipitation method[J]. Journal of Lanzhou Jiaotong University, 2018, 37(5): 80-84. doi: 10.3969/j.issn.1001-4373.2018.05.014 [10] 王国建, 王东田, 陈霞, 等. 吸附法除氟技术的原理与方法[J]. 环境科学与管理, 2008, 33(8): 121-124, 165. doi: 10.3969/j.issn.1673-1212.2008.08.034Wang Guojian, Wang Dongtian, Chen Xia, et al. Mechanism and method of fluoride removal by adsorption[J]. Environmental Science and Management, 2008, 33(8): 121-124, 165. doi: 10.3969/j.issn.1673-1212.2008.08.034 [11] Samarghandi M R, Khiadani M, Foroughi M, et al. Defluoridation of water using activated alumina in presence of natural organic matter via response surface methodology[J]. Environmental Science and Pollution Research, 2016, 23(1): 887-897. doi: 10.1007/s11356-015-5293-x [12] 左思敏, 荆肇乾, 陶梦妮, 等. 天然沸石和改性沸石在废水处理中的应用研究[J]. 应用化工, 2019, 48(5): 1136-1139, 1145. doi: 10.3969/j.issn.1671-3206.2019.05.035Zuo Simin, Jing Zhaoqian, Tao Mengni, et al. Application of natural zeolite and modified zeolite in wastewater treatment[J]. Applied Chemical Industry, 2019, 48(5): 1136-1139, 1145. doi: 10.3969/j.issn.1671-3206.2019.05.035 [13] 张启伟, 王桂仙. 竹炭对饮用水中氟离子的吸附条件研究[J]. 广东微量元素科学, 2005, 12(3): 63-66. doi: 10.3969/j.issn.1006-446X.2005.03.016Zhang Qiwei, Wang Guixian. Research on removing effects of the bamboo-carbon for fluoride from drink water[J]. Trace Elements Science, 2005, 12(3): 63-66. doi: 10.3969/j.issn.1006-446X.2005.03.016 [14] 张瑞玲, 李德生, 秦松岩, 等. 新型生物质炭吸附剂对地下水中氟的去除作用[J]. 现代食品科技, 2014, 30(10): 159-164. https://www.cnki.com.cn/Article/CJFDTOTAL-GZSP201410027.htmZhang Ruiling, Li Desheng, Qin Songyan, et al. Removal of fluoride from groundwater using new biochar adsorbents[J]. Modern Food Science and Technology, 2014, 30(10): 159-164. https://www.cnki.com.cn/Article/CJFDTOTAL-GZSP201410027.htm [15] Kim M, Choong C E, Hyun S, et al. Mechanism of simultaneous removal of aluminum and fluoride from aqueous solution by La/Mg/Si-activated carbon[J]. Chemosphere, 2020, 253: 126580. doi: 10.1016/j.chemosphere.2020.126580 [16] 严云, 张宇豪, 曹芮, 等. 磁性荞麦壳炭的制备对水中Cr(Ⅵ)吸附性能的研究[J]. 环境科学与技术, 2019, 42(1): 8-14. https://www.cnki.com.cn/Article/CJFDTOTAL-FJKS201901002.htmYan Yun, Zhang Yuhao, Cao Rui, et al. Study of magnetic buckwheat hull biochar: preparation and Cr(Ⅵ) adsorption characteristics[J]. Environmental Science & Technology, 2019, 42(1): 8-14. https://www.cnki.com.cn/Article/CJFDTOTAL-FJKS201901002.htm [17] Liang Jian, Cai Qin, Li Bo, et al. Preparation and adsorption properties of activated carbon from glutinous rice straw by KOH method[J]. Agricultural Science & Technology, 2015, 16(11): 2549-2551, 2567. http://qikan.cqvip.com/Qikan/Article/Detail?id=666795512 [18] 侯嫔, 岳烨, 张犇, 等. 荞麦壳基活性炭的制备及其性能研究[J]. 矿业科学学报, 2020, 5 (1): 122-130. https://www.cnki.com.cn/Article/CJFDTOTAL-KYKX202001013.htmHou Pin, Yue Ye, Zhang Ben, et al. Preparation and characterization of Buckwheat shell-based activated carbon[J]. Journal of Mining Science and Technology, 2020, 5(1): 122-130. https://www.cnki.com.cn/Article/CJFDTOTAL-KYKX202001013.htm [19] 陈秋娟, 谢微, 莫福旺, 等. 磷酸法荔枝核基活性炭的制备及其吸附性能[J]. 食品工业, 2020, 41(11): 62-65. https://www.cnki.com.cn/Article/CJFDTOTAL-SPGY202011015.htmChen Qiujuan, Xie Wei, Mo Fuwang, et al. Preparation and adsorption properties of lychee kernel activated carbon by phosphoric acid method[J]. The Food Industry, 2020, 41(11): 62-65. https://www.cnki.com.cn/Article/CJFDTOTAL-SPGY202011015.htm [20] 张会均, 欧阳晚秋, 陈琳莉. 磷酸法茶渣活性炭的制备及吸附性能[J]. 应用化工, 2019, 48(12): 2947-2950. https://www.cnki.com.cn/Article/CJFDTOTAL-SXHG201912034.htmZhang Huijun, Ouyang Wanqiu, Chen Linli. Characteristics and optimization of activated carbon from tea waste using H3PO4[J]. Applied Chemical Industry, 2019, 48(12): 2947-2950. https://www.cnki.com.cn/Article/CJFDTOTAL-SXHG201912034.htm [21] 邢宝林, 谌伦建, 张传祥, 等. KOH活化法制备褐煤基活性炭的活化机理研究[J]. 中国矿业大学学报, 2014, 43(6): 1038-1045. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGKD201406013.htmXing Baolin, Chen Lunjian, Zhang Chuanxiang, et al. Activation mechanism oflignite-based activated carbon prepared by KOH activation[J]. Journal of China University of Mining & Technology, 2014, 43(6): 1038-1045. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGKD201406013.htm [22] 孙龙梅, 张丽平, 薛建华, 等. 活性炭制备方法及应用的研究进展[J]. 化学与生物工程, 2016, 33(3): 5-8. doi: 10.3969/j.issn.1672-5425.2016.03.002Sun Longmei, Zhang Liping, Xue Jianhua, et al. Research progress on preparation methods and applications of activated carbon[J]. Chemistry & Bioengineering, 2016, 33(3): 5-8. doi: 10.3969/j.issn.1672-5425.2016.03.002 [23] 左宋林. 磷酸活化法制备活性炭综述(Ⅰ)——磷酸的作用机理[J]. 林产化学与工业, 2017, 37(3): 1-9. doi: 10.3969/j.issn.0253-2417.2017.03.001Zuo Songlin. Review on phosphoric acid activation for preparation of activated carbon(Ⅰ): roles of phosphoric acid[J]. Chemistry and Industry of Forest Products, 2017, 37(3): 1-9. doi: 10.3969/j.issn.0253-2417.2017.03.001 [24] 范世锁, 刘文浦, 王锦涛, 等. 茶渣生物炭制备及其对溶液中四环素的去除特性[J]. 环境科学, 2020, 41(3): 1308-1318. https://www.cnki.com.cn/Article/CJFDTOTAL-HJKZ202003036.htmFan Shisuo, Liu Wenpu, Wang Jintao, et al. Preparation of tea waste biochar and its application in tetracycline removal from aqueous solution[J]. Environmental Science, 2020, 41(3): 1308-1318. https://www.cnki.com.cn/Article/CJFDTOTAL-HJKZ202003036.htm [25] Vithanage M, Herath I, Joseph S, et al. Interaction of arsenic with biochar in soil and water: a critical review[J]. Carbon, 2017, 113: 219-230. doi: 10.1016/j.carbon.2016.11.032 [26] 刘凌言, 陈双荣, 宋雪燕, 等. 生物炭吸附水中磷酸盐的研究进展[J]. 环境工程, 2020, 38(11): 91-97. https://www.cnki.com.cn/Article/CJFDTOTAL-HJGC202011015.htmLiu Lingyan, Chen Shuangrong, Song Xueyan, et al. Research progress in removal of phosphate from water by biochar[J]. Environmental Engineering, 2020, 38(11): 91-97. https://www.cnki.com.cn/Article/CJFDTOTAL-HJGC202011015.htm [27] 王雪颖, 徐冰峰, 喻岚, 等. 咖啡渣颗粒活性炭制备及应用研究进展[J]. 净水技术, 2020, 39(8): 80-88, 108. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSJS202008017.htmWang Xueying, Xu Bingfeng, Yu Lan, et al. Research progress on preparation and application of granular activated carbon from coffee grounds[J]. Water Purification Technology, 2020, 39(8): 80-88, 108. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSJS202008017.htm [28] 黄循精. 咖啡副产品的化学成分与综合利用[J]. 热带作物研究, 1987, 7(4): 68-70. https://www.cnki.com.cn/Article/CJFDTOTAL-RDNK198704019.htmHuang Xunjing. Chemical composition and comprehensive utilization of coffee by products[J]. Chinese Journal of Tropical Agriculture, 1987, 7(4): 68-70. https://www.cnki.com.cn/Article/CJFDTOTAL-RDNK198704019.htm [29] 庄圣炜, 孙水裕. 咖啡渣制备活性炭及其在环境污染治理应用进展[J]. 广东化工, 2018, 45(11): 110-113. doi: 10.3969/j.issn.1007-1865.2018.11.048Zhuang Shengwei, Sun Shuiyu. Preparation of activated carbon made from coffer grounds and its application in the environmental pollution control: a review[J]. Guangdong Chemical Industry, 2018, 45(11): 110-113. doi: 10.3969/j.issn.1007-1865.2018.11.048 [30] Hou P, Cannon F S, Nieto-Delgado C, et al. Effect of preparation protocol on anchoring quaternary ammonium/epoxide-forming compound into granular activated carbon for perchlorate adsorption: Enhancement by response surface methodology[J]. Chemical Engineering Journal, 2013, 223(3): 309-317. http://www.sciencedirect.com/science/article/pii/S1385894713003306 [31] 苑守瑞, 朱义年, 梁美娜. 氯化锌活化法制备柚子皮活性炭[J]. 环境科学与技术, 2010, 33(S1): 22-25. https://www.cnki.com.cn/Article/CJFDTOTAL-FJKS2010S1008.htmYuan Shourui, Zhu Yinian, Liang Meina. Preparation of activated carbon from grapefruit bran by activation with zinc chloride[J]. Environmental Science & Technology, 2010, 33(S1): 22-25. https://www.cnki.com.cn/Article/CJFDTOTAL-FJKS2010S1008.htm [32] 郭刚军, 马尚玄, 胡小静, 等. 氯化锌活化制备澳洲坚果壳活性炭试验[J]. 林业工程学报, 2020, 5(6): 106-113. https://www.cnki.com.cn/Article/CJFDTOTAL-LKKF202006015.htmGuo Gangjun, Ma Shangxuan, Hu Xiaojing, et al. Preparation of the activated carbon from Macadamia shell by zinc chloride activation[J]. Journal of Forestry Engineering, 2020, 5(6): 106-113. https://www.cnki.com.cn/Article/CJFDTOTAL-LKKF202006015.htm [33] 王云波, 谭万春, 王晓昌, 等. 沸石、骨炭、活性氧化铝除氟效果研究[J]. 西安建筑科技大学学报: 自然科学版, 2002, 34(4): 325-328. doi: 10.3969/j.issn.1006-7930.2002.04.006Wang Yunbo, Tan Wanchun, Wang Xiaochang, et al. A study on the effect of fluoride removal by activated zeolite, activated alumina and bone char[J]. Journal of Xi'an University of Architecture & Technology, 2002, 34(4): 325-328. doi: 10.3969/j.issn.1006-7930.2002.04.006 [34] 谢虹, 贾文波, 吴志刚. 活性氧化铝除氟剂的除氟性能研究[J]. 华中科技大学学报: 医学版, 2005, 34(5): 644-646. doi: 10.3870/j.issn.1672-0741.2005.05.041Xie Hong, Jia Wenbo, Wu Zhigang. Performance of activated aluminum oxide as a fluorine-removal agent[J]. Acta Universitatis Medictnae Tangji, 2005, 34(5): 644-646. . doi: 10.3870/j.issn.1672-0741.2005.05.041 [35] Mullick A, Neogi S. Synthesis of potential biosorbent from used stevia leaves and its application for malachite green removal from aqueous solution: kinetics, isotherm and regeneration studies[J]. RSC Adv, 2016, 6(70): 65960-65975. doi: 10.1039/C6RA15225B [36] Mullick A, Neogi S. Ultrasound assisted synthesis of Mg-Mn-Zr impregnated activated carbon for effective fluoride adsorption from water[J]. Ultrasonics Sonochemistry, 2019, 50: 126-137. doi: 10.1016/j.ultsonch.2018.09.010 -
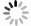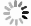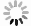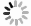





 下载:
下载: